From Research to Manuscript: Considering Scientific Writing in Institutional Contexts
Scientific writing lies at the core of research communication, serving to present results, enhance institutional credibility, satisfy regulatory requirements, and disseminate knowledge to both professional and public audiences. Building on themes presented in an Asian Council of Science Editors webinar from Research to Manuscript: The Role of Scientific Writing in Institutional Contexts, this article explores how writing underpins research, teaching, and communication. Key qualities of effective scientific writing are identified, as are ethical principles and collaboration strategies. Common challenges such as authorship disputes, missed deadlines, and unclear responsibilities are discussed. The authors explain how plain language and the responsible use of artificial intelligence can strengthen scientific communication. Practical guidance, including authorship frameworks and process flowcharts are presented. Collectively, these insights emphasize that scientific writing is not merely a technical task but a professional and ethical responsibility that shapes the visibility, credibility, and trustworthiness of science.
| Copyright © 2026 DeTora et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Research achieves its full significance only when shared with the scientific community and the broader public, allowing others to evaluate, validate, and build upon it. Scientific writing serves as the primary medium through which experiments, observations, and analyses are transformed into accessible knowledge1-3. Within institutional settings, writing extends beyond communication: It helps establish credibility, supports scholarly and institutional profiles, and serves as a marker of professional identity4. Universities, companies, governments, and publishers rely on well-prepared scientific writing to document studies, meet regulatory requirements, and guide decision-making5. The diversity of audiences and formats, however, presents challenges for both authors and reviewers6.
Scholars such as linguist John Swales describe the shared practices of writing as forming “discourse communities”. A discourse community may include researchers within a field, authors publishing in the same journal, or smaller groups such as members of an academic department7. These communities share
values regarding research credibility, ethical principles, and effective communication practices. Various external bodies, government agencies, journal editors, and industry guideline committees, such as the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), provide directions to help authors meet the expectations of specific audiences8. By adhering to these established conventions, researchers do not merely record results; they participate in communities that ensure continuity, coherence, and integrity in science9.
This article explores how scientific writing functions within institutions, highlighting the qualities that make it effective and the ethical and collaborative practices that support its reliability. A comprehensive review of existing literature demonstrates how prior studies have addressed institutional scientific writing, identifies gaps, and situates the present work within the broader scholarly context10,11. The authors discuss the professional, ethical, and communicative responsibilities that accompany effective scientific writing.
ROLE OF SCIENTIFIC WRITING IN INSTITUTIONAL CONTEXTS
Overview: Scientific writing creates an authoritative record of study design, data collection, and analysis, ensuring that findings are verifiable, reproducible, and extendable through peer review. It establishes transparency and accountability within the research process, reinforcing institutional credibility and scientific rigor. Beyond its descriptive function, scientific writing also operates as a persuasive medium that facilitates research funding and policy influence. Well-structured proposals, progress reports, and publications help researchers secure grants and shape research programs. International bodies such as the International Committee of Medical Journal Editors (ICMJE), the International Council for Harmonisation (ICH), and the Council of Science Editors (CSE) underscore the importance of ethical preparation, accurate reporting, and clarity in presenting scientific data.
Scientific writing also has an essential pedagogical role. Within academic institutions, it cultivates critical thinking and precision in communication through structured assignments such as lab reports, literature reviews, and research articles. Researchers must employ persuasive arguments in grant applications, project evaluations, and conference presentations, key venues for advancing institutional visibility and scholarly discourse.
Scientific writing shapes policy and public discourse: In broader contexts, scientific writing extends beyond academia to shape policy, clinical practice, and technological innovation. It informs guidelines, editorials, and policy briefs that influence public health, environmental governance, and engineering advancement. Thus, it bridges scientific evidence with societal application, ensuring that research outcomes contribute meaningfully to decision-making processes.
Scientific writing is not merely expository; it inherently integrates rhetorical strategies that frame the significance of a topic, identify knowledge gaps, and interpret results persuasively. According to John Swales’ Creating a Research Space (CARS) model, research article introductions typically progress through three rhetorical stages:
| • | Establishing a territory | |
| • | Identifying a niche | |
| • | Occupying the niche |
These rhetorical moves guide readers toward recognizing the novelty, relevance, and scholarly contribution.
| Table 1: | Functions of scientific writing in institutional contexts | |||
| Function | Example outputs | Institutional purpose |
| Research | Protocols, manuscripts, articles, reports, grant proposals, IRB/EC applications |
Planning for research, obtaining ethical reviews, seeking funding, dissemination of data, archival records |
| Teaching | Lab reports, reviews, reflections, presentation materials | Training and evaluation |
| Communication | Guidelines, policy briefs, editorials | Facilitating regulatory compliance, reputation building, public trust |
Functions of scientific writing: The functions of scientific writing often intersect across institutional boundaries. For instance, a clinical trial report may fulfill both a regulatory obligation and a scholarly publication requirement. Similarly, clinical protocols serve not only to guide study conduct but also to stand as publishable research outputs. In addition, datasets derived from such studies are frequently shared through public repositories, enhancing transparency and accessibility.
Recognizing these overlapping roles enables institutions and authors to leverage scientific writing as a tool for accountability, ethical compliance, and global knowledge exchange.
Table 1 summarizes the key functions of scientific writing within institutions, linking each function to typical outputs and the purposes they serve. Writing supports research by producing protocols, manuscripts, reports, grant proposals, and ethical review applications, which are essential for planning studies, obtaining approvals, securing funding, disseminating findings, and maintaining archival records. In the context of teaching, outputs such as lab reports, reviews, reflections, and presentation materials help train students, evaluate learning outcomes, and foster critical thinking. For communication, documents like guidelines, policy briefs, and editorials enable institutions to comply with regulations, build reputation, and maintain public trust. Together, these examples illustrate that scientific writing is a multifunctional tool integral to research, education, and knowledge dissemination within institutional frameworks.
Qualities of effective scientific writing: Not all writing is equally effective. Three qualities are particularly important when considering scientific writing.
Scientific rigor: A paper must be built on sound methods, reliable data, and careful analysis. Without rigor, the writing cannot carry weight, no matter how polished the language may be. Rigor is assured by careful and ethical study design, scrupulous documentation at every stage of the project, and proper use of scientific and statistical methods to ensure unbiased communication of results. The peer review process is meant to maintain that rigor; despite the current critiques of this process, it remains the only reliable means to date to ensure objectivity and quality in scientific publications.
Adherence to conventions: Scientific fields rely on shared formats so that readers know what to expect. Following established standards, such as those of the International Committee of Medical Journal Editors (ICMJE) or the Council of Science Editors (CSE), ensures that manuscripts meet professional norms12,13. In addition to documentation and formatting standards, scientific genres also need to conform to established rules. For example, the scientific research article, the primary unit of conveying scientific research results, needs to include an informative title, abstract, and keywords, and at least 4 standard sections: Introduction, Methods, Research, and Discussion (IMRaD), followed sometimes by a conclusion, as well as author disclosures and bibliography. Review papers can be narrative or systematic; the latter need to conform to the PRISMA statement for systematic reviews (https://www.prisma-statement.org/). Abstracts and grant proposals come with their own guidelines and rules depending on the publication venue or funding agency. These standards and rules must be observed at the inception stage of a scientific writing project.
Clarity: Research should be understandable on the first reading. Applying plain language principles, using clear sentences, complete tables and figures, and simple explanations where possible, does not reduce scientific value but makes the work stronger and more widely accessible14,15. Clarity implies that every effort is made to minimize and ideally eliminate ambiguity (which can happen at word, sentence, paragraph, or idea level); sometimes, an unclear sentence is the result of unclear thinking. At the heart of scientific writing is precision, with its close companion, brevity.
Audience awareness: Underlying all these principles is the overarching rhetorical concept of audience. Given its goals, scientific writing needs to adapt to its audience, who demand rigor, clarity, and precision, as well as adherence to conventions, to expedite understanding. Furthermore, different journals have different audiences with diverse interests or knowledge bases; similarly, grant proposal panels might consist of people with diverse levels of expertise, including lay reviewers such as patients. Authors should familiarize themselves with the type of work published by journals or funded by agencies and understand how to target their audience(s).
ETHICAL CONSIDERATIONS IN SCIENTIFIC WRITING
Writing cannot be separated from ethics, and scientific writing, especially, must reflect the basic ethical principles inherent to the research field for the studies being reported. A recent survey by the Asian Council of Science Editors, by Maryam Sayab and colleagues, highlighted how institutional pressures contribute to unethical practices. More than one-third of respondents reported pressure to compromise research integrity, while over half believed that institutional publication requirements promote guest authorship or predatory practices16. These findings underscore how systemic incentives shape the challenges of scientific writing.
At the research stage, ethics ensures that studies respect the basic rights of their participants, use sound methods, and maintain integrity, guided by frameworks such as the Declaration of Helsinki or Good Clinical Practice17. In general, scientific and biomedical journals require that any research reported meets the ethical requirements relevant to the field. The ICMJE, for instance, provides Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals12. These include basic guidelines for ensuring that reported research meets basic ethical standards as well as guaranteeing that authors disclose all relevant financial and nonfinancial interests. The ICMJE also includes similar guidance for editors and peer reviewers, emphasizing the need for ethics across the writing and publishing process.
Within an individual work, ethics requires transparency, honesty in data presentation, and proper recognition of contributions17. Publication ethics extends these responsibilities further, not only to authors but also to reviewers and editors, who share accountability for ensuring integrity, fairness, and rigor in the scholarly record. Authors must avoid plagiarism, declare conflicts of interest, acknowledge funding sources, avoid redundant or duplicate publications, and increasingly disclose the use of artificial intelligence18. Reviewers and editors are likewise expected to uphold confidentiality, provide unbiased evaluations, and identify ethical concerns such as data falsification, inappropriate image manipulation, or breaches in research standards.
Ethical research also involves adherence to discipline-specific standards, including proper care and use of animals, human subjects’ protections, and responsible data management. Moreover, visual representations, figures, graphs, and data displays must be designed to convey information truthfully and accessibly, reflecting principles of clarity and integrity as emphasized by scholars such as Edward Tufte.
Guidelines such as Good Publication Practice for Company-Sponsored Research (GPP 2022) provide frameworks for multidisciplinary teams to collaborate responsibly while aligning with the ICMJE Recommendations, relevant laws, and ethical principles that recognize the shared responsibility of all participants in the publication process19.
| Table 2: | Ethical considerations in scientific writing | |||
| Domain | Key principles | Institutional standards |
| Research ethics | Data integrity, participant protection | Declaration of Helsinki, GCP, GLP |
| Writing ethics | Transparency, fair credit, honesty | ICMJE, CSE guidelines |
| Publication ethics | Avoid plagiarism, disclose conflicts and AI | COPE, GPP guidelines |
The use of artificial intelligence also raises ethical questions about confidentiality, authorship, and transparency. Current best practice is to avoid entering sensitive data into open AI systems and to disclose all AI use openly, as recommended by GPP 2022 and ICMJE guidelines18.
At the same time, the scholarly publishing landscape faces broader integrity challenges. The proliferation of predatory journals, often lacking rigorous peer review and editorial oversight, undermines trust and visibility in legitimate research. Compounding this are the ongoing replication and peer review crises, as well as an excessive reliance on citation-based metrics such as journal impact factors, which can distort research priorities and assessment practices. Addressing these interconnected issues requires strengthening transparency, quality control, and responsible evaluation across the entire publication ecosystem.
Table 2 outlines the key ethical domains relevant to scientific writing, highlighting the associated principles and institutional or international standards that guide responsible practice. In research ethics, maintaining data integrity and protecting participants are central, with frameworks such as the Declaration of Helsinki, Good Clinical Practice (GCP), and Good Laboratory Practice (GLP) providing guidance. Writing ethics emphasizes transparency, fair attribution of credit, and honesty, with standards outlined by the International Committee of Medical Journal Editors (ICMJE) and the Council of Science Editors (CSE). Publication ethics focuses on avoiding plagiarism, disclosing conflicts of interest, and the responsible use of artificial intelligence, with adherence to guidelines from the Committee on Publication Ethics (COPE) and Good Publication Practice (GPP). This table demonstrates that ethical scientific writing spans the entire research and publication process, ensuring credibility, accountability, and trustworthiness within institutional and scholarly communities.
COLLABORATION IN SCIENTIFIC WRITING
Most manuscripts today are written by teams, not individuals. This makes collaboration a skill as important as writing itself. In a paper outlining important skill sets for medical writers, Clemow and colleagues emphasize teamwork and effective listening as well as the ability to build consensus as essential to producing high-quality documents20. The GPP 2022 also emphasizes the need for effective teamwork among authoring groups, especially practices such as defining roles and responsibilities in writing, allowing authors ample opportunity to comment on drafts, and establishing means of resolving potential differences of opinion before they occur.
Successful collaboration begins with clarity: Authors should be identified early, and roles assigned using tools like the CRediT taxonomy (Fig. 1), which specifies contributions such as conceptualization, data curation, or drafting19. Of note, the CRediT roles emphasize scientific research settings and may not cover all applicable research tasks in clinical studies or work in health economics.
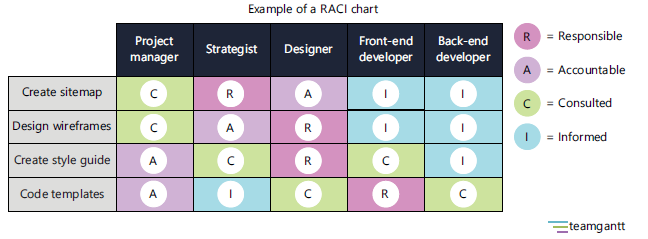
The RACI diagram (Fig. 2), identifies a specific person responsible, accountable, consulted, and informed for each critical task in completing a writing project, and can also help by mapping who does what in order to avoid redundancies and prevent omissions20.
During training events such as the ISMPP-ACSE joint webinar on GPP 2022, the speakers emphasized that defining roles and responsibilities early in the writing process not only clarifies accountability but also strengthens ethical practices21. It is always easier for researchers and authors to meet clearly defined ethical responsibilities in the context of specific tasks.

|
|
Of course, effective collaboration requires more than frameworks. Teams must set realistic timelines, circulate drafts with enough time for review, and send clear and specific reminders to keep work on track. Disagreements are inevitable, but open communication and adherence to shared guidelines can usually resolve conflicts without damaging trust22. Additional practices, such as identifying a specific decision-maker to resolve scientific or procedural questions, can reduce the burden on team members by creating clear lines of responsibility and accountability.
Authors writing within institutions often encounter unexpected obstacles. Deadlines may be missed, for instance, or miscommunications can occur. Simple steps like scheduling writing time in advance, blocking calendars, and using automatic reminders can help support team members with busy schedules and allow them to meet timelines. Planning writing around competing commitments also helps to set realistic goals. Drafts and comments produced in a rush may be inaccurate or incomplete, which can cause confusion and communication gaps. Drafts that are fragmented or inconsistent may reflect poor coordination or possibly a lack of time for one or more authors. Regular meetings and active listening can help restore alignment, but early and realistic planning is also needed.
(Planning→Drafting→Collaboration→Review→Submission→Dissemination)
|
Authorship disputes are common and can be damaging to personal and professional relationships. The ICMJE and CSE both provide guidelines for authorship these are similar but not fully aligned. Therefore, as suggested in GPP 2022, it is important to create early agreement regarding authorship, based on the guidelines for the target journal. The GPP 2022 also provides advice for choosing authors in the context of very large studies through the use of steering committees. Another difficulty arises when different journals or agencies impose specific requirements regarding authorship that are not fully aligned with ICMJE or CSE standards; sharing links to these guidelines early can help teams adapt quickly or may encourage further discussion about the choice of journal.
Another problem may arise in overlooking the acknowledgment of important contributions to a work or in confusion over whether a contribution is only acknowledged or confers authorship. These problems are best prevented by setting authorship criteria at the outset and using recognized standards such as CRediT to identify the work that should be acknowledged23.
Institutional and global perspectives: New challenges in scientific writing are constantly emerging. Writing for the public or for policy makers requires plain language and the involvement of non-specialist reviewers (Fig. 3).
Writing is shaped not only by institutional expectations but also by global forces, expectations among different communities of researchers, journal editors, and fellow researchers. Early-career researchers may experience a struggle to balance institutional and other writing conventions with a personal style, but most scholarly writing in the sciences requires consistency with a specific journal style, aligned with writing by other experts in a scientific field, rather than individuality. Opportunities for personal voice, such as editorials or commentaries, often remain limited compared to the demands of formal manuscripts. Younger researcher, however, may be able to produce work in social media that comments on their publications, allowing for a balance of personal expression with the scientific formality of journals.
For scientists in non-English-speaking contexts, barriers to clarity and visibility remain significant. Even strong research may fail to reach a wide audience if the manuscript lacks fluency. Institutions can support their researchers by offering editing assistance, translation services, and budgeting for publication fees. More broadly, journals and indexing bodies should recognize the need to value content quality over linguistic polish or geographic origin, helping reduce systemic inequities in scholarly publishing24-26.
CONCLUSION
Scientific writing is not merely the final step of research; it can be a defining stage that shapes how institutions demonstrate accountability, uphold ethics, and share discoveries with the world. Effective writing integrates rigor, clarity, adherence to disciplinary conventions, and ethical integrity. It is inherently collaborative and influenced by both institutional frameworks and global standards.
Although challenges remain, such as authorship disputes and the responsible use of AI, these can be addressed through careful planning, transparent communication, and robust institutional support. By embracing these practices, researchers can produce writing that is accurate, accessible, trustworthy, and impactful. Ultimately, scientific writing is both a professional responsibility and a vital contribution to the collective credibility and visibility of science.
SIGNIFICANCE STATEMENT
Scientific writing is a cornerstone of research, teaching, and institutional communication, yet its role is often underestimated. This article highlights how well-structured writing enhances credibility, supports ethical and collaborative practices, and facilitates knowledge dissemination across professional and public audiences. By linking writing outputs to institutional purposes and emphasizing ethical standards, the authors underscore that scientific writing is not merely a technical task but a professional and ethical responsibility. As such, writing is critical for maintaining the visibility, trustworthiness, and impact of science.
REFERENCES
- Busse, C.E., E.W. Anderson, T. Endale, Y.R. Smith, M. Kaniecki, C. Shannon and E.T. August, 2022. Strengthening research capacity: A systematic review of manuscript writing and publishing interventions for researchers in low-income and middle-income countries. BMJ Global Health, 7.
- Ryba, R., Z.A. Doubleday, M.J. Dry, C. Semmler and S.D. Connell, 2021. Better writing in scientific publications builds reader confidence and understanding. Front. Psychol., 12.
- Norjuliyati Binti Hamzah, S.R.B.C. Balian, N. Akmaliah Binti Dzulkurnain, N. Abbasvandi and N.A. Adrina Binti Daud, 2024. An investigation on scientific writing difficulties & writing process. Int. J. Acad. Res. Bus. Social Sci., 14: 1608-1620.
- Sánchez, M.J.F. and M.T.B. Traver, 2020. Study of the scientific writing strategies used by novice researchers. Braz. J. Educ. Technol. Soc., 13: 329-337.
- Costley, J., K. Adamovich, G. Shulgina, M. Baldwin and M. Fanguy, 2025. Computer-mediated collaborative writing and individual scientific writing proficiency. J. Engl. Acad. Purposes, 76.
- Giuffredi, R., V. Grasso and A. L’Astorina, 2024. Web-based science communication at research institute level: Balancing dissemination, dialogue and promotion in a major Italian scientific institution. Front. Commun., 9.
- Hussain, M., R. Rehman and M. Baig, 2020. Manuscript writing and publication workshop: An invoking pilot study on enhancing cognitive research capabilities in health sciences institutes of Pakistan. Cureus, 12.
- Gopee, N. and M. Deane, 2013. Strategies for successful academic writing-institutional and non-institutional support for students. Nurse Educ. Today, 33: 1624-1631.
- Rasool, T. and M. Dilshad, 2023. Factors influencing research culture at public universities in Pakistan. Res. J. Societal Issues, 5: 41-58.
- Swales, J.M., 2016. Reflections on the concept of discourse community. ASp, 69: 7-19.
- Holstein, S.E., K.R.M. Steinmetz and J.D. Miles, 2015. Teaching science writing in an introductory lab course. J. Undergrad. Neurosci. Educ., 13: A101-A109.
- ICMJE, 2024. Recommendations for the conduct, reporting, editing, and publication of scholarly work in medical journals.
- CSE, 2023. Recommendations for Promoting Integrity in Scientific Journal Publications. Council of Science Editors, Mullica Hill, New Jersey, Pages: 93.
- Barroga, E. and G.J. Matanguihan, 2021. Creating logical flow when writing scientific articles. J. Korean Med. Sci., 36.
- Plain Language Europe, 2022. Plain Language Principles.
- Sayab, M., L.M. deTora and M. Sarwar, 2025. Publication pressure vs research integrity: Global insights from an Asian Council of Science Editors survey. Sci. Editor, 48.
- ICH, 2016. Efficacy guidelines.
- Gasparyan, A.Y., M. Yessirkepov, A.A. Voronov, S.V. Gorin, A.M. Koroleva and G.D. Kitas, 2016. Statement on publication ethics for editors and publishers. J. Korean Med. Sci., 31: 1351-1354.
- DeTora, L.M., D. Toroser, A. Sykes, C. Vanderlinden and F.J. Plunkett et al., 2022. Good publication practice (GPP) guidelines for company-sponsored biomedical research: 2022 update. Ann. Internal Med., 175: 1298-1304.
- Muller, B.J., B.S. Cade and L. Schwarzkopf, 2018. Effects of environmental variables on invasive amphibian activity: Using model selection on quantiles for counts. Ecosphere, 9.
- Clemow, D.B., B. Wagner, C. Marshallsay, D. Benau and D. L’Heureux et al., 2018. Medical writing competency model-section 2: Knowledge, skills, abilities, and behaviors. Ther. Innovation Regul. Sci., 52: 78-88.
- National Information Standards Organization, 2005. CRediT, Contributor Roles Taxonomy. NISO, USA, ISBN: 9781950980185, Pages: 12.
- Lingard, L., 2021. Collaborative writing: Strategies and activities for writing productively together. Perspect. Med. Educ., 10: 163-166.
- Minor, S. and S. Stumbar, 2022. Writing collaboratively in medical education. Med. Sci. Educ., 32: 517-522.
- Andrade, E.L., A.F. Bento, J. Cavalli, S.K. Oliveira and R.C. Schwanke et al., 2016. Non-clinical studies in the process of new drug development-Part II: Good laboratory practice, metabolism, pharmacokinetics, safety and dose translation to clinical studies. Braz. J. Med. Biol. Res., 49.
- Sarwar, M., D. Toroser, L. de Tora, L. Dormer and S.T. Mathew, 2023. Promoting GPP 2022: ACSE and ISMPP joint webinar. Trends Scholarly Publ., 2: 18-20.
How to Cite this paper?
APA-7 Style
DeTora,
L., Hanganu-Bresch,
C., Sarwar,
M. (2026). From Research to Manuscript: Considering Scientific Writing in Institutional Contexts. Trends in Scholarly Publishing, 5(1), 28-36. https://doi.org/10.21124/tsp.2026.28.36
ACS Style
DeTora,
L.; Hanganu-Bresch,
C.; Sarwar,
M. From Research to Manuscript: Considering Scientific Writing in Institutional Contexts. Trends Schol. Pub 2026, 5, 28-36. https://doi.org/10.21124/tsp.2026.28.36
AMA Style
DeTora
L, Hanganu-Bresch
C, Sarwar
M. From Research to Manuscript: Considering Scientific Writing in Institutional Contexts. Trends in Scholarly Publishing. 2026; 5(1): 28-36. https://doi.org/10.21124/tsp.2026.28.36
Chicago/Turabian Style
DeTora, Lisa, Cristina Hanganu-Bresch, and Muhammad Sarwar.
2026. "From Research to Manuscript: Considering Scientific Writing in Institutional Contexts" Trends in Scholarly Publishing 5, no. 1: 28-36. https://doi.org/10.21124/tsp.2026.28.36

This work is licensed under a Creative Commons Attribution 4.0 International License.




